The RegenLab BCT family kits are designed to prepare Autologous - Platelet Rich Plasma from a small sample oft he patients own blood. Each BCT tube draws 10cc of venous blood and yields an average of 5.5cc of autologous platelet concentrate. RegenLab USA offers procedure kits consisting 1, 2 or 4 BCT processing tubes. Which kit to use is to be determined by the physicians required volume of PRP. The RegenKit BCT 1, 2 and 4 consist of all blood draw and transfer accessories. The RegenKit BCT 3 contains 3 processing tubes only, individually sterile packed.

What is A-PRP® / PRP™?
- A-PRP®/ PRP™ is the patient's platelet concentrate prepared with RegenKit®
- The physician draws a small amount of venous blood and isolates the red cells from a platelet concentrate, embedded in its own plasma. The entire process takes less then 10 minutes.
- As A-PRP® is processed from the patients own blood, there is very little risk of allergy or rejection with this treatment.
A-PRP® / PRP™ Components
Platelets are key factors in hard and soft tissue repair mechanisms. They provide the essential growth factors FGF, PDGF, TGF-ß, EGF, VEGF, IGF that are involved in stem cell migration, differentiation and proliferation. The stimulation of fibroblasts and endothelial cells induces new extracellular matrix deposition and neo-vascularization.
Plasma is essential for cell survival as it contains nutrients, vitamins, hormones, electrolytes and proteins. Proteins are key molecules for the coagulation process and the formation of the fibrin polymer that will serve as a scaffold for cell migration, differentiation and proliferation.
Optimal Platelet Concentration
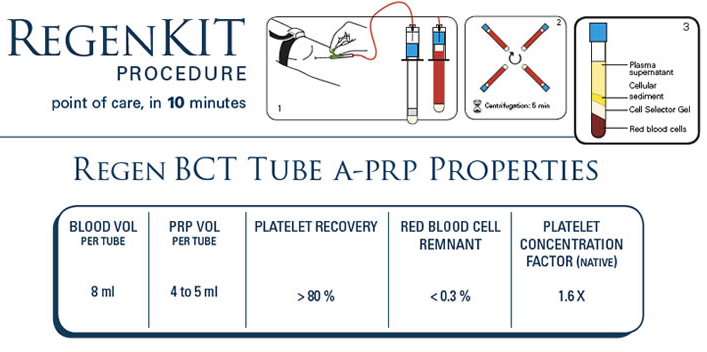
The Regen™ BCT (Blood Cell Therapy) tube prepares an average of 5.5cc of autologous platelet rich plasma with a platelet recovery superior to 80% and a concentration factor of 1.6-fold. Although the system is technically capable of producing significantly higher platelet concentrations, it is not what research shows to be beneficial for clinical use. More and more studies demonstrate that concentrations of platelets 1 to 3 times over the baseline show more robust healing rates than those with concentrations of 3 to 8 times the baseline.
Some studies even showed that too high platelet concentrations may actually have negative effects. In a in vivo study it was shown that highly concentrated platelet preparations had an inhibitory influence on osteoblast activity, probably due to unwanted inhibitory and cytotoxic effects of growth factors at such high concentrations. Similarly an in vitro study, demonstrated that platelet concentration over 2.5-fold, resulted in a reduction in proliferation and a suboptimal effect on osteoblast function.
White Blood Cells in Tissue Regeneration
White blood cells are strongly reduced (> 85% depletion) in Regen BCT A-PRP®.
The role of white blood cells (WBC) in healing is controversially discussed among physicians. To address this concern scientifically, WBC subsets, granulocytes and mononuclear cells, need to be looked at individually. Granulocytes, and more specifically neutrophils, are the front-line defenders against invading pathogens and are associated with the inflammatory response. They release a large variety of highly active antimicrobial substances and proteases. Uncontrolled release of these factors can cause severe damage to the tissue, delay healing rates and increase the risk of scarring. Thanks to the RegenKit BCT technology more than 96% of the granulocytes are removed from A-PRP®.
The few white blood cells still present in A-PRP® are mostly mononuclear cells (lymphocytes and monocytes). These two cell types are also involved in the immune response but have been shown to support the healing process.
West Coast Laser & Aesthetics is committed to helping physicians build successful aesthetic practices. Our laser experts will help you determine the right technologies for your patients' needs and assist you in developing your practice.
Email: contact@westcoastlaser.com
Phone: 503-291-6959
Fax: 503-291-6960
7824 SW 37th Ave. Ste. C
Portland, OR 97225
© 2020 West Coast Laser & Aesthetics
© 2024 West Coast Laser



